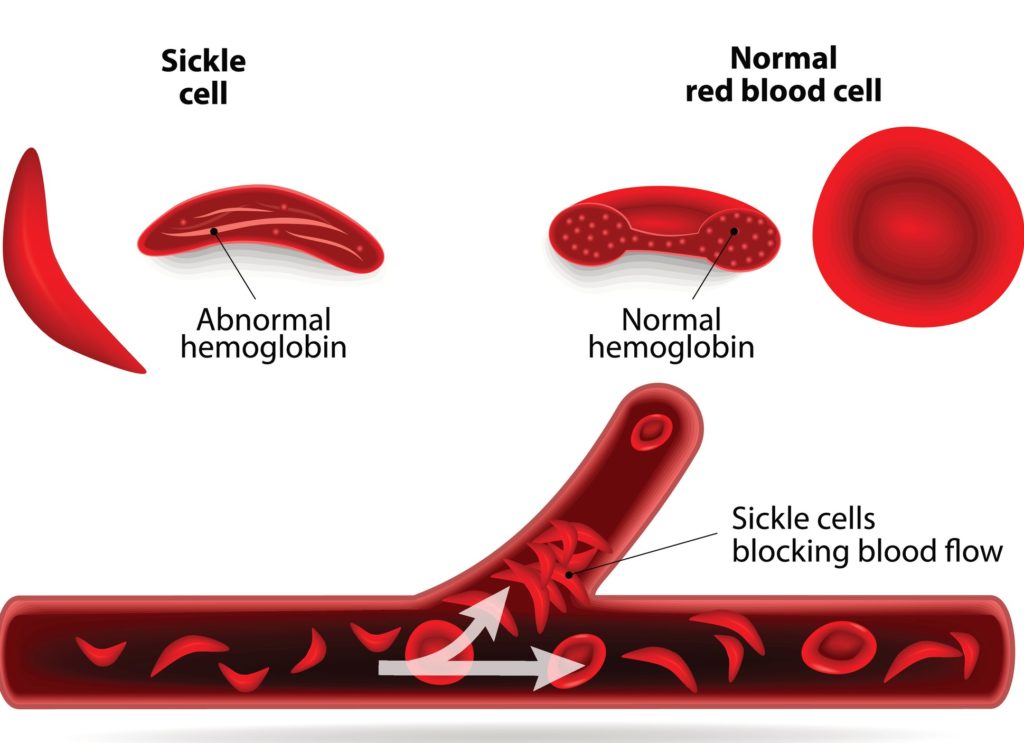
For the first time in 20 years, the FDA has approved two transformative new treatments for the approximately 100,000 people with sickle cell disease (SCD). One new med will prevent the excruciating pain of SCD while the other is formulated to prevent organ damage.
“After decades of waiting, there is now a treatment option that could change the course of this disease,” Beverley Francis-Gibson, president and CEO of the Sickle Cell Disease Association of America, said in a news release. “Hope has never been higher for people living with sickle cell disease with a pipeline of new treatments on the horizon.
The opportunity before us in the coming months and years is profound and historic because, in most cases, medications treat disease complications or symptoms but not the condition itself. Finally, these two new medications the U.S. Food and Drug Administration (FDA) recently approved for Sickle Cell Disease are vastly different in that respect.”
Adakveo (crizanlizumab)
Manufactured by Novartis, Adakveo can prevent vaso-occlusive episodes of unbearable pain that occur when malformed blood cells get caught in blood vessels. Vaso-occlusive crisis is extremely painful and is a frequent reason for emergency department visits and hospitalization for patients with sickle cell disease.
Adakveo is administered by infusion once per month and you must be at least 16 years to be approved to use. The side effects are moderate including nausea, back or joint pain and fever. Infusion dosage depends on patient’s weight.
Oxbryta (voxelotor)
Developed by Global Blood Therapeutics, Oxbryta is an