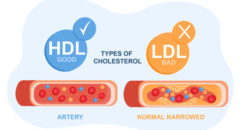
Could a popular pain reliever put you at a greater risk of heart attack and stroke? This could be the case considering the fact that the U.S. Food and Drug Administration (FDA) recently issued an updated warning label for popular pain relievers – NSAIDs (nonsteroidal anti-inflammatory drugs) such as ibuprofen, naproxen and Celebrex. NSAIDs are also used in cold, flu and sleep medications. The FDA is set to make similar warning changes to lower-dose over-the-counter (OTC) NSAIDs, including Aleve (the least likely to increase the risk of heart attacks and strokes), Motrin and Advil as well.
Could a popular pain reliever put you at a greater risk of heart attack and stroke? This could be the case considering the fact that the U.S. Food and Drug Administration (FDA) recently issued an updated warning label for popular pain relievers – NSAIDs (nonsteroidal anti-inflammatory drugs) such as ibuprofen, naproxen and Celebrex. NSAIDs are also used in cold, flu and sleep medications. The FDA is set to make similar warning changes to lower-dose over-the-counter (OTC) NSAIDs, including Aleve (the least likely to increase the risk of heart attacks and strokes), Motrin and Advil as well.
The OTC drugs in this group are used for the temporary relief of pain and fever. The prescription drugs in this group are used to treat several kinds of arthritis and other painful conditions. The FDA mentions that although aspirin is also an NSAID, this revised warning doesn’t apply to aspirin or Tylenol.