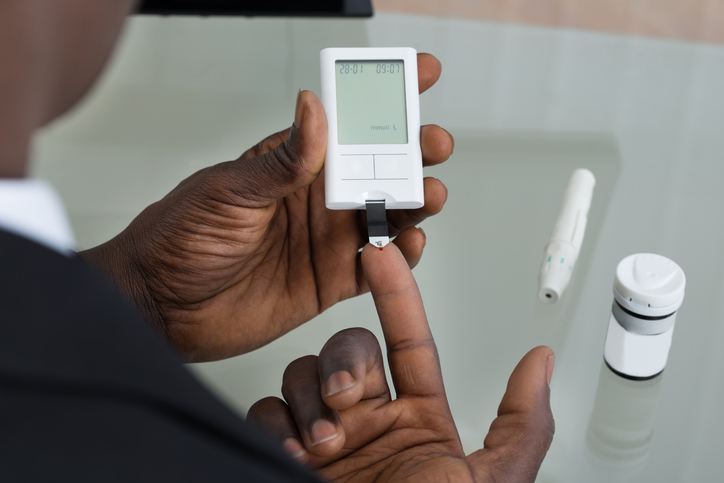
Here’s a bit of good news for diabetics: the fingerstick test could be a thing of the past.
The U.S. Food and Drug Administration recently decided that the Dexcom G5 continuous monitoring system can help replace the finger-stick method in determining insulin dosages.
Previously, the monitoring system was used as a complement to fingersticking for diabetes treatment. The Dexcom G5 is the first FDA-approved continuous monitoring system that can be used without fingersticking.
“The FDA works hard to help ensure that novel technologies, which can reduce the burden of daily disease management, are safe and accurate,” said Alberto Gutierrez, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
The system, however, still requires calibration with two fingersticks each day, Gutierrez adds. But it does eliminate the need for fingerstick testing to make treatment decisions.







